FEI Prohibited Substances: The 2026 Digital Compliance Guide

In 2026, Clean Sport under the Fédération Équestre Internationale (FEI) is no longer just about avoiding obvious doping violations. It’s about precision, documentation, and digital accountability.
Consultants working with international riders and veterinary teams have observed that many professionals assume good intentions are enough. They aren’t. The FEI operates under the doctrine of Strict Liability: if a prohibited substance is found in your horse, the responsibility lies with you—regardless of intent.
The reality on the ground is this: modern laboratories using high-sensitivity Mass Spectrometry can detect substances at nanogram levels. Compliance in 2026 is forensic-level science. For a deeper understanding of how these scientific standards affect physical performance, see EquiTests 2026: Precision Pattern Mastery via Kinematic Spatial Mapping
The FEI Regulatory Structure (EADCMP) — What You’re Really Managing
Under the FEI’s Equine Anti-Doping and Controlled Medication Programme (EADCMP), substances fall into two primary categories:
- Banned Substances
- Controlled Medication
Understanding the difference isn’t academic — it determines the trajectory of your case if something goes wrong.
Banned vs. Controlled Substances — 2026 Technical Comparison
Under the regulatory authority of the Fédération Équestre Internationale (FEI) and its Equine Anti-Doping and Controlled Medication Programme (EADCMP), the classification below determines both scientific interpretation and legal exposure.
| Category | Definition & Intended Use | Clinical Examples & Trade Names | Legal Consequences – 2026 Context |
|---|---|---|---|
| Banned Substances | Substances associated with performance manipulation or doping. No legitimate therapeutic justification during competition. Presence indicates prohibited enhancement or major rule violation. | Anabolic Steroids (Stanozolol), Erythropoietin (EPO), Clenbuterol (at high concentrations) | Classified as Major Violations. Up to 4 years suspension for a first-time major violation, fines exceeding $15,000 USD, disqualification of results, forfeiture of medals/prize money, and potential legal costs. Reputational damage and sponsorship termination are common secondary consequences. |
| Controlled Medication | Substances used for legitimate veterinary therapy, including NSAIDs and anti-inflammatories. Permitted for treatment but must be fully cleared prior to competition. | Phenylbutazone (Bute), Flunixin (Banamine), Dexamethasone | Classified as Controlled Medication violations. Automatic disqualification of results, fines typically $1,500–$5,000 USD, possible short-term suspension, and mandatory recording of the case in the FEI HorseApp and regulatory database. Increased scrutiny in subsequent events is standard. |
Professional Compliance Alert
This table serves as a technical summary for the 2026 season. Under the Strict Liability principle, the Person Responsible (PR) is solely accountable for any prohibited substance found in a horse’s sample. Regulations and FEI Sanctions change frequently. Always verify specific substances via the Official FEI Database before administration.
Technical Compliance Notes (2026)
- The term Sanctions is not generic. The FEI now distinguishes sharply between Major (Banned) and Controlled Medication cases.
- Detection thresholds in 2026 are lower due to high-sensitivity analytical methods.
- Even therapeutic administration of Bute or Banamine without appropriate withdrawal strategy can trigger regulatory action.
- Clenbuterol cases increasingly hinge on concentration levels and pharmacokinetic interpretation.
In practical compliance management, the category dictates not only medical planning but legal risk modeling.
Detection Times vs. Withdrawal Periods — A Forensic Risk Analysis (2026)
Speaking plainly as a forensic pharmacologist: this is where most otherwise careful riders get into serious legal trouble.
The confusion between Detection Time and Withdrawal Period is not academic — it is regulatory exposure measured in months of suspension and five-figure fines.
Detection Time: A Laboratory Statistic
A Detection Time is the period during which a substance or its metabolites can be identified in a biological sample using accredited laboratory equipment — specifically ultra-high sensitivity tandem Mass Spectrometry.
In 2026, this matters more than ever.
Modern labs can detect trace nanogram concentrations that were simply invisible in 2018. The analytical floor has dropped. Substances linger at detectable levels long after clinical effect appears gone.
This is what I call the 2026 Detection Trap.
Riders relying on older “safe” timing assumptions are operating with outdated margins.
Why Individual Physiology Changes the Equation
Published detection times are generated from controlled studies using limited sample groups. Your horse is not an average.
Small physiological variations can shift a detection window by 12–24 hours — sometimes more:
- Hydration levels influence renal clearance. A slightly dehydrated horse eliminates drugs more slowly.
- Body fat percentage affects lipophilic drug storage. Fat-soluble compounds redistribute and release over time.
- Training intensity alters circulation and metabolic rate.
- Stress and travel can modify elimination kinetics.
This is applied Pharmacokinetics, not theory.
And under Strict Liability, variability is your problem — not the laboratory’s.
Withdrawal Period: A Strategic Buffer, Not a Scientific Endpoint
A Withdrawal Period is not the same thing as a Detection Time.
- Detection Time = laboratory-derived statistical average.
- Withdrawal Period = strategic safety buffer designed to reduce risk.
The FEI publishes Detection Times as guidance. They are derived from structured studies with limited cohorts and standardized conditions.
They are not a legal defense.
Official reference:
👉 https://inside.fei.org/fei/cleansport/detection-times
If a laboratory identifies a prohibited substance, citing a published average will not shield you from sanctions.
The 2026 Safety Margin Doctrine
In today’s testing environment, conservative modeling is mandatory.
Because Mass Spectrometry sensitivity has increased, statistical averages now carry higher risk. A detection window that once had comfortable margin may now sit dangerously close to competition day.
As a rule:
- Never plan to the minimum detection threshold.
- Add a safety buffer based on travel stress, event timing, and individual horse variables.
- Document every calculation.
I routinely advise teams to extend withdrawal buffers beyond published averages when competition stakes are high.
Digital Safeguards: Remove Memory from the Process
Human memory is the shortest path to a $15,000 fine.
In 2026, there is no justification for relying on handwritten notebooks or mental timelines.
Use Digital Medication Logbooks such as:
- BarnManager
- Equilab
These systems automate:
- Timestamped administration logs
- Withdrawal countdown calculations
- Compliance history records
- Audit-ready documentation trails
When integrated correctly, they create defensible timelines aligned with modern enforcement standards.
Practical Legal Reality
If your horse tests positive:
- The laboratory will reference analytical confirmation via tandem Mass Spectrometry.
- The FEI will apply the Strict Liability doctrine.
- The burden of explanation shifts to you.
Detection Times are scientific guidance.
Withdrawal Periods are strategic tools.
Neither replaces conservative judgment.
In 2026, the safest rider is the one who assumes detection capability exceeds the published average — and plans accordingly.
The FEI CleanSport App — Your First Legal Shield (2026)
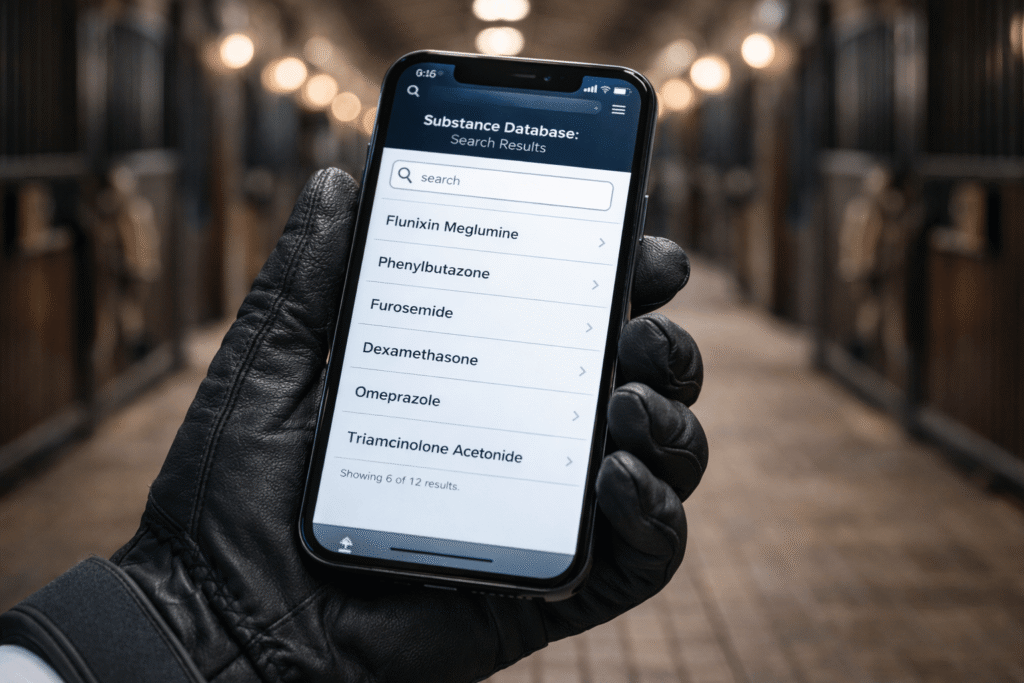
If you do nothing else in your compliance workflow, do this: check the FEI CleanSport App before any substance enters your horse.
The official Clean Sport portal is here:
👉 https://inside.fei.org/fei/cleansport
In 2026, this isn’t a convenience tool. It’s your primary due-diligence instrument.
The app allows you to:
- Search by trade name
- Search by active ingredient
- Confirm classification (Banned vs. Controlled)
- Cross-check regulatory notes instantly
From a tribunal perspective, “I didn’t know” is irrelevant. Under the authority of the Fédération Équestre Internationale (FEI), demonstrating that you checked the official database before administration is often the first evidentiary step in arguing No Fault or No Significant Negligence.
In 2026, the FEI HorseApp — mandatory for health and temperature logging at international events — should be synchronized with your internal medication logs. When these systems align, they form what regulators increasingly expect:
A Defensible Digital Audit Trail.
Your smartphone is no longer a convenience device. It is compliance infrastructure.
Digital Medication Logbooks — Metadata Is Evidence

Paper notebooks are operationally obsolete.
In a 2026 regulatory dispute, what matters is not just what you recorded — but how and when you recorded it.
Apps such as BarnManager and Equilab provide:
- Timestamped entries
- User attribution (who logged the treatment)
- GPS-tagged metadata
- Cloud-backed storage
- Audit-ready export capability
This is called Metadata Evidence.
A handwritten note can be questioned.
A time-stamped, system-locked digital entry is far harder to dismantle.
Withdrawal Countdown Calculators
One of the most protective features in modern logbook apps is the Withdrawal Countdown Calculator.
Instead of estimating clearance windows manually, the system:
- Logs the exact administration time
- Applies your configured safety buffer
- Displays a competition-clearance date
Human calculation errors are one of the most common triggers of Controlled Medication violations.
Technology removes arithmetic from the risk equation.
And in 2026, arithmetic mistakes are expensive.
The Caffeine Trap — A No-Fluff Reality Check
Caffeine and Theobromine are Threshold Substances.
That means small levels are tolerated below a defined limit — but once exceeded, the result becomes a violation.
In my experience, most caffeine positives are not caused by a dramatic mistake. They stem from Cumulative Environmental Failure.
Examples include:
- Poor feed room hygiene
- Shared scoops between supplements and human food
- Cocoa-shell bedding
- Unsealed feed bins
A single drop of coffee is rarely the issue.
Repeated low-level exposure is.
Human-to-Horse Transfer (2026 Forensic Risk)
There is another overlooked pathway: topical NSAIDs.
Products containing diclofenac (e.g., Voltaren-type gels) applied to a groom’s hands can transfer:
- From hand
- To feed bucket
- To bit
- To the horse’s mouth
Modern laboratories can identify trace compounds via high-sensitivity analysis. Environmental transfer is not hypothetical — it is documented.
Under Strict Liability, source explanation does not eliminate responsibility.
Pro-Tip: Digital Stable Hygiene
- No human food in feed rooms.
- Dedicated supplement scoops.
- Gloves when handling topical NSAIDs.
- Log feed batch numbers digitally.
- Verify supplements through: https://informed-sport.com
- Cross-check every substance in the FEI CleanSport Portal before use.
The 2026 Compliance Reality
- FEI CleanSport App → substance verification
- Digital Medication Logbook → metadata evidence
- FEI HorseApp → official event synchronization
Today, compliance is not paperwork. It is systems integration.
Together, they create defensible documentation.
When a laboratory report lands on a tribunal desk, your smartphone records may determine whether the narrative is negligence — or disciplined risk management.
In 2026, your most important piece of safety equipment isn’t hanging in the tack room.
The 2026 Rapid Response Protocol
(When Medication Is Administered Close to Competition)
In a regulated FEI environment, hesitation is risk. The moment treatment is given, documentation must begin.
Step 1: Verify Immediately in the FEI CleanSport Tools
Search both the trade name and active ingredient inside the official Clean Sport database. Confirm classification (Banned vs. Controlled) and review any regulatory notes.
If competing internationally, ensure the treatment is aligned with requirements under the Fédération Équestre Internationale (FEI).
Step 2: Log Comprehensive Metadata Evidence
Record the following in your Digital Medication Logbook:
- Exact date and time
- Dosage and route of administration
- Treating veterinarian
- Batch number
- Photograph of the product label
- Expiry date
This is not administrative overkill. It is evidentiary protection.
Immediately synchronize relevant health records inside the FEI HorseApp if required for competition monitoring. In 2026, fragmented records weaken your defensibility.
Step 3: Calculate a Conservative Safety Buffer
Use a Withdrawal Countdown Calculator inside your logbook system.
Do not rely solely on published detection averages. Add a buffer based on:
- Travel stress
- Hydration variability
- Body condition
- Event timing
Human memory and mental math are the shortest path to sanctions.
Digital systems reduce that exposure.
Laboratory Reality (2026): What You’re Up Against

Modern FEI-accredited laboratories operate with High-Sensitivity Tandem Mass Spectrometry capable of identifying trace nanogram concentrations.
What has changed since 2018 is not just sensitivity — it is pattern recognition.
In 2026, laboratories also deploy Big Data Anomaly Detection. This means:
- Longitudinal biological data is reviewed
- Repeated micro-level findings are flagged
- Patterns resembling pharmacological manipulation are investigated
Even if a substance is detected at a low concentration, the broader analytical profile may trigger scrutiny.
This is no longer simple substance identification.
It is statistical behavioral analysis at scale.
If a case proceeds to tribunal, analytical confirmation via tandem Mass Spectrometry becomes the evidentiary backbone.
Your defense rests on documentation integrity — not assumptions about low concentration levels.
Frequently Asked Questions (2026 Compliance)
1. Can caffeine really cause a positive test?
Yes — but context matters.
Caffeine and Theobromine are Threshold Substances, meaning they are permitted below defined concentration limits. Exceeding that threshold — often due to cumulative environmental exposure — triggers a violation.
Feed contamination, cocoa-shell bedding, and repeated minor exposure create risk.
2. Can human medications cause a violation?
Yes.
Topical NSAIDs containing Diclofenac (e.g., Voltaren-type gels) present a documented forensic transfer risk. Residue from hands can contaminate feed buckets, bits, or stable surfaces.
Under Strict Liability, source explanation does not remove responsibility.
3. Are published withdrawal times legally protective?
No.
Detection Times are laboratory-derived averages. Withdrawal periods are strategic buffers. Neither guarantees clearance.
Only conservative planning, supported by defensible digital records, reduces risk.
Performance Engineering & Compliance Integration
Regulatory safety and biomechanical performance planning must operate together.
Medication timing influences recovery cycles, tissue loading, and competition readiness. Forward-thinking teams integrate compliance modeling with movement analytics.
For a deeper technical framework, review our core resource:
👉 Explore the EquiTests 2026 Biomechanics Manual — Align Performance Data with Regulatory Strategy
The most successful international programs in 2026 treat compliance as part of performance architecture.
Because at this level, regulatory discipline is not separate from competitive excellence.
All data regarding substance compliance and user queries is handled according to the EquiApps Equestrian Data & Privacy Policy to ensure total rider confidentiality.